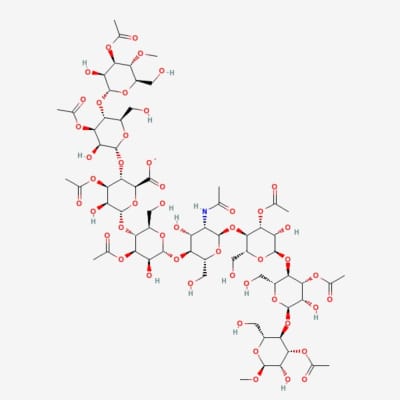
Acemannan (ACE) is a polysaccharide (carbohydrate) located within the soft pulp of the inner leaves of Aloe plants. Many of the beneficial properties of Aloe have been attributed to the acemannan present in the gel.
Composition
Acemannan is a β(1,4)-acetylated mannan-based polysaccharide derived from the plant aloe vera (Barbadensis milleri).
Healing Properties
Acemannan has potential immunostimulant, antiviral, antineoplastic, and gastrointestinal properties.
Anti-Aphthous activity
Helps prevent/treat aphthous ulcers (small, shallow sores inside the mouth or at the base of the gums).[1]
Antiviral
Antineoplastic
Helps prevent, inhibit or halt the development of a neoplasm (a tumor).[3]
Bone Health
(Skeletal Health)
Osteogenic
Promotes the functioning of osteogenesis (i.e. producing bone)
Bone Regeneration
Acemannan, a β-(1–4)-acetylated polymannose extracted from Aloe vera gel, has been proposed as biomaterial for bone regeneration.[3:1]
- Acemannan is an effective bioactive agent for bone regeneration, enhancing bone growth as assayed in two- and three-dimensions.[3:2]
- Treatment with Acemannan for calvarial (skullcap, or the upper part of the neurocranium which covers the cranial cavity containing the brain) defect healing showed a significant increase in bone surface and bone volume and tissue mineral density. The acemannan-treated groups also had a denser bone matrix.[3:3]
Gastrointestinal Health
Immunostimulant
Oral Health
Disease / Symptom Treatment
Diabetes
Acemannan is degraded by the intestinal microbiota to form oligosaccharides. These oligosaccharides inhibit intestinal glucose absorption.[4]
Glucose Regulation
Acemannan treatment promotes a significant reduction in blood glucose.[4:1]
Blood Pressure
Acemannan treatment promotes a significant reduction in blood pressure.[4:2]
Lipid Profile Regulation
Acemannan treatment promotes improvement of the lipid profile.[4:3]
Viral Infections
HIV
Title: Medicinal plant and their bioactive phytochemicals in the treatment of recurrent aphthous ulcers: A review of clinical trials
Publication: Pharmacognosy Reviews
Date: May 2018
Study Type: Human Study: Review of clinical trials
Author(s): Fatemeh Heydarpour, Masoomeh Abasabadi, Zahra Shahpiri, Siavash Vaziri, Hesam Aldin Nazari, Fariba Najafi, Maryam Mirzaei, Mohammad Hosein Farzaei
Institution(s): Kermanshah University of Medical Sciences, Kermanshah, Iran; Tehran University of Medical Sciences, Tehran, Iran; Tabriz University of Medical Science, Tabriz, Iran
Copy: archive, archive-mirror ↩︎Title: Enhancement of allo-resposiveness of human lymphocytes by acemannan (CarrisynTM)
Publication: International Journal of Immunopharmacology
Date: March 1988
Study Type: Human Study: In Vitro
Author(s): Debra Womble, J. Harold Helderman
Institution(s): The University of Texas Southwestern Medical Center at Dallas, USA
Copy: archive, archive-mirror ↩︎Title: Acemannan increased bone surface, bone volume, and bone density in a calvarial defect model in skeletally-mature rats
Publication: Journal of Dental Sciences
Date: July 2018
Study Type: Animal Study: In Vivo
Author(s): Dyna Jeanne D. Godoya, Jaroenporn Chokboribal, Ruben Pauwels, Wijit Banlunara, Polkit Sangvanich, Sukanya Jaroenporn, Pasutha Thunyakitpisal
Institutions: Chulalongkorn University, Bangkok, Thailand; Phranakhon Rajabhat University, Bangkok, Thailand; Biomedical Sciences Group, KU Leuven, Belgium
Copy: archive, archive-mirror ↩︎ ↩︎ ↩︎ ↩︎Title: Compositional and Structural Features of the Main Bioactive Polysaccharides Present in the Aloe vera Plant
Publication: Journal of AOAC International
Date: November 2019
Study Type: Review of Plant Composition
Author(s): Minjares-Fuentes R, Femenia A, Comas-Serra F, Rodríguez-González VM
Institution(s): Universidad Juárez del Estado de Durango, Durango, México; University of the Balearic Islands, Palma de Mallorca, Spain
Copy: archive, archive-mirror ↩︎ ↩︎ ↩︎ ↩︎